Malaysia Medical Device Regulation 2012 mandated establishments playing the role as an local authorized representative for medical device, importer and distributor establish certify with the Good Distribution Practice for Medical Device (GDPMD) with the objective to ensure that all medical devices distributed in the country are handled, stored, transported, and traced under safe and controlled conditions, safeguarding patient safety and regulatory compliance.
GDPMD specifies the requirements for a good distribution system to be established, implemented and maintained by an establishment in carrying out activities in the medical device supply-chain to comply with Malaysian medical device regulatory requirements as stipulated in Medical Device Act 2012 (Act 737). GDPMD requires an establishment to demonstrate its ability to maintain quality, safety and performance of medical devices in compliance with the Malaysian medical device regulatory requirement throughout the supply-chain. It shall be used by both the internal and external parties to determine the ability of an establishment to meet the requirements specified within.
The practicality and sustainability of a GDPMD system depend heavily on how well the system is designed. A poorly designed system can create unnecessary burdens, disrupt medical device distribution operations, and compromise compliance with applicable requirements—ultimately leading to significant operational challenges.
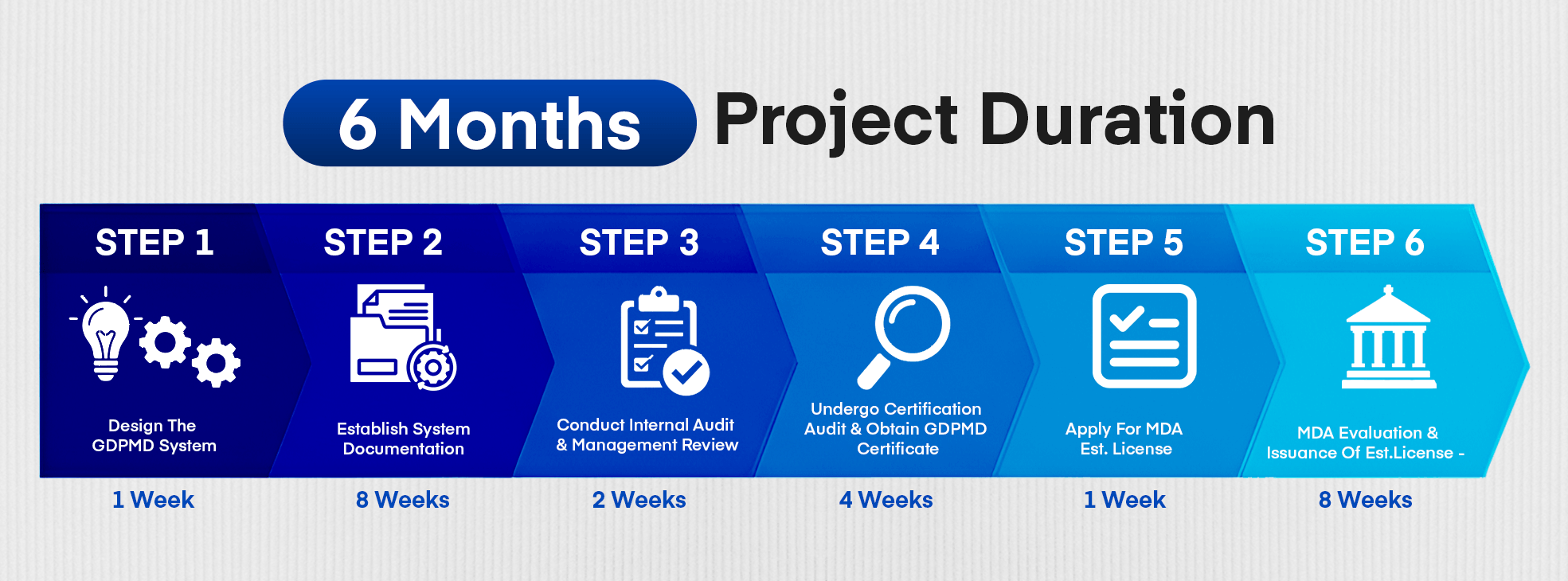
Step 1 – Design the GDPMD System
Develop the framework and structure of the Good Distribution Practice for Medical Devices (GDPMD) system tailored to your organization’s operations.
Step 2 – Establish System Documentation
Prepare and formalize all required policies, procedures, manuals, and records to ensure compliance with GDPMD standards.
Step 3 – Conduct Internal Audit and Management Review
Conduct an internal audit to verify system compliance and effectiveness, followed by a management review to evaluate compliance, assess effectiveness, consider changes, identify improvement opportunities, and establish an action plan.
Step 4 – Undergo Certification Audit & Obtain GDPMD Certificate
Engage an accredited CAB to conduct the certification audit of your GDPMD system. Receive the official GDPMD certificate upon successful completion of the audit.
Step 5 – Apply for MDA Establishment License
Submit the establishment license application to the MDA along with the GDPMD certificate and supporting documents.
Step 6 – MDA Evaluation & Issuance of Establishment License
The MDA reviews and evaluates the application for compliance with regulatory requirements. Upon approval, the MDA issues the establishment license, authorizing your organization to operate in accordance with the laws.
Get Expert GDPMD Compliance Support Today
With years of experience and a highly competent team, our GDPMD consultants provide reliable support to help your business stay compliant with Malaysian medical device regulatory requirements. From system setup to certification and license application, we manage the entire process so you can focus on growing your business with confidence.





